Assisted Reproduction For Male Infertility
Assisted Reproduction Techniques

Infertility affects 8 million couples in the U.S. In roughly 50% of couples, there is an associated male issue or “factor.” Here we discuss how assisted reproduction can be used to treat (more accurately “bypass”) the problem of male infertility. Since the birth of Louise Brown in 1978, assisted reproductive techniques (ART) are now widely used, with more than 8 million children conceived worldwide with ART to date. Currently, in the U.S., 2% of all babies born annually are IVF babies.
Dr. Paul Turek of the Turek Clinic has played a vital role in the development of these technologies to help infertile men. As a male fertility specialist, he uses the full range of ART to help his patients conceive. After thoroughly examining the patient’s reproductive potential, he can recommend the most effective treatment and help to grow their families.
Intrauterine insemination (IUI)

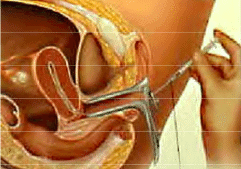
Intrauterine insemination (IUI) involves the mechanical placement of a washed pellet of ejaculated sperm within the female uterus, beyond the cervix, with a soft catheter. The placement of sperm with IUI is deeper in the female reproductive tract than sperm are normally “placed” with intercourse (i.e., the upper vagina).
This procedure is timed to take place during the “fertile” or “ovulatory” window in the woman’s menstrual cycle. If a woman has cervical issues, using IUI to bypass the cervix may improve the chances for successful conception.
IUI also addresses several male infertility issues — specifically, problems caused by:
- Low sperm counts
- Low sperm motility
- Immunologic infertility due to antisperm antibodies
- Mechanical problems with sperm delivery (such as hypospadias, erectile dysfunction or ejaculation disorders)
To be successful, there should be at least 5 to 40 million motile (moving) sperm in the ejaculate (volume x concentration x motility) and at least 5 million motile sperm should be inseminated after the ejaculate is processed. Success rates vary widely and are generally related to female reproductive potential, with pregnancy rates per cycle (month) ranging from 1 to 15%. If these target numbers for processed sperm are met, then pregnancy rates with IUI are most closely linked to female age and reproductive potential.
In cases of male factor infertility, overall pregnancy rates with IUI are generally improved if the ovaries are stimulated with medication to make more than one egg available every month (also termed ovarian stimulation).
In Vito Fertilization (IVF)

IVF is a medical procedure performed on women that involves:
- Ovarian stimulation
- Egg retrieval
- Fertilization of the egg in the laboratory
- Embryo transfer to the uterus
The goal of hormonal stimulation during IVF is to develop more than one mature follicle so that enough eggs are retrieved later to counteract the inherent inefficiencies of fertilization as well as the limitations of placing sperm and eggs together in the laboratory. For IVF, the female menstrual cycle is highly regulated in what is termed controlled ovarian stimulation.
The majority of the stimulation protocols currently used start by down-regulating the pituitary gland to prevent spontaneous hormonal activity. Subsequently, pituitary hormones are replaced with gonadotropins (FSH and LH) each day for 9 to 13 days and ovarian follicle development is carefully monitored with ultrasound. Each follicle contains an individual maturing egg or oocyte. Once adequate follicular size is achieved, another injection is administered to induce ovulation, and eggs are retrieved and collected surgically 36 hours later, right before they would naturally ovulate. This is performed transvaginally without an incision and guided by ultrasound. All eggs retrieved are then nurtured in the embryology laboratory.
A semen sample, generally obtained by masturbation on the same day as egg retrieval, is also needed for IVF. The semen is washed so that the sperm is separated from the semen fluid and the sperm are incubated with the eggs at a density of at least 100,000 sperm/egg.
After 12 to 18 hours of co-incubation in an incubator, the eggs are observed to see if they have fertilized. With normal fertilization, 2 pronuclei are present and there is an extrusion of the second polar body. The embryos are allowed to divide in the incubator for up to 6 or 7 days and are graded for quality based on the number of cells present and the shape of the embryo as it develops. The reason for in vitro culture of embryos for up to a week is that the hardier embryos will survive beyond the first several cell divisions. There is an increased chance of implantation and pregnancy with hardier embryos if they are cultured for 5-7 days after retrieval. Because they are more robust, fewer blastocysts are transferred to the uterus (day 5-7) than earlier stage embryos (around day 3) to obtain similar pregnancy rates. This also decreases the miscarriage rate and reduces the chance of having twins or triplets. However, because the laboratory culture conditions do not perfectly replicate the natural environment of the uterus, some experts believe that some perfectly healthy embryos may not survive the weeklong laboratory incubation process and therefore believe that an earlier transfer (day 3) of embryos is warranted in some cases.
It has also become clear that pregnancy rates with IVF improve if embryos are transferred back into a normal, hormonally unstimulated uterus. To accomplish this, developing embryos are frozen on days 5-7 of culture, instead of transferring them back into the uterus immediately. In these cases, embryo transfer is done during an ovulatory cycle after the IVF cycle, when the uterine environment returns to its more “normal” state.
During embryo transfer the embryo is transferred back into the uterus in a small procedure without anesthesia. The embryos are loaded into a soft catheter with a small volume of culture and then expelled into the uterus with a syringe. The number of embryos transferred back depends on several factors, including the age of the patient and the quality of the embryos, because the “take” rate or implantation rate is not 100% for every embryo returned. The popularity of SET or “single embryo transfers” is currently extremely high worldwide and supported by national guidelines, as this dramatically reduces the rate of having multiple (i.e., twins, triplets or higher) gestations with IVF.
The indications for the use of IVF for male infertility are listed in the Table.

Intracytoplasmic Sperm Injection (ICSI)
In 1992, intracytoplasmic sperm injection (ICSI), a laboratory addition or “add on” to IVF, was invented. With this technique, individual sperm are pulled by suction into a microscopic pipette after crushing the sperm tails to immobilize them. The sperm are injected directly through the eggshell (zona pellucida) covering the unfertilized egg through the (perivitelline) space that separates the egg from the shell, and directly into the egg itself.
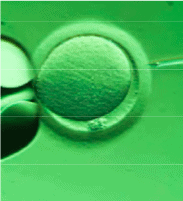
With this technique, the numerical sperm requirement for egg fertilization dropped from hundreds of thousands of moving sperm for each egg with IVF to a single live sperm for ICSI. This approach allows “immature” sperm obtained surgically or by aspiration from men with no sperm in the ejaculate to be used for conception.
Indeed, ICSI has become so popular that over 75% of IVF cases performed in US clinics now routinely use this technique to boost fertilization rates during IVF. In fact, the ability of ICSI to improve egg fertilization with live sperm from any source in the man has encouraged some fertility experts to look beyond the ejaculate and into the male reproductive tract to find sperm for biologic pregnancies. At present, sources of sperm that are amendable for use with ICSI include sperm obtained from the vas deferens, epididymis, and testicle.

Sperm Morphology
Sperm are considered normal or abnormal based on a list of specific features and measurements of the head, midpiece and sperm tail. An increased percentage of abnormal sperm within a semen sample is termed teratozoospermia. Sperm morphology may correlate with fertility potential in that it may affect:
- Sperm’s ability to successfully penetrate the cervix
- Sperm’s ability to bind to the egg “shell” or zona pellucida
- The embryo implantation rate
In cases of low sperm morphology, ICSI is done with IVF and appears to “rescue” many cases that may have resulted in failed fertilization with IVF alone.
Success Rates for Assisted Reproductive Technology (ART) Procedures
Fertility centers report pregnancy outcomes in a complex matter. The proportion of eggs that fertilize in a dish with 2 pronuclei within 18 hours of sperm insemination, termed the normal fertilization rate, is a commonly used, early (but indirect) measure of ART success. More importantly, however, are the following accepted definitions of pregnancy outcomes with ART:
- Live birth/cycle: The percentage of ART cycles started that result in a live birth. This rate is the most meaningful outcome measure from ART procedures.
- Pregnancy/cycle: The percentage of ART cycles started that produce a pregnancy. This rate is higher than the live birth per cycle rate because some pregnancies end in miscarriage, induced abortion, or stillbirth.
- Live birth/egg retrieval: The percentage of ART cycles in which eggs are retrieved that result in a live birth. It is generally higher than the live birth/cycle rate because it excludes cycles that were canceled before eggs were retrieved.
- Live birth/transfer rate: Includes only those ART cycles in which an embryo or egg and sperm were transferred back to the woman. This rate is the highest of the measures of ART success.
- Singleton live birth/cycle: The percentage of ART cycles started that result in a singleton live birth. This has become a very important ART outcome measure, as singleton births have a much lower risk than multiple infant births for adverse infant health outcomes, including prematurity, low birth weight, disability, and death.
Risks and Complications of ART Procedures
As a medical procedure, IVF can have complications that are medication related, procedure related, or procedure associated. These types of complications are outlined below.
- Medication-related complications: These include ovarian hyperstimulation syndrome (OHSS) and ovarian torsion. Thought to be a result of excessive hormone stimulation, severe OHSS occurs in <1% of IVF cycles but can be life threatening and is closely monitored, often in a hospital setting, until it resolves. Ovarian torsion is also rare and involves the twisting of the ovary on its blood supply. It is due to the large increase in the size of the ovary with hormone stimulation and is treated surgically by untwisting the ovary.
- Procedure-related complications: The process of egg retrieval and embryo transfer to the uterus have low complication rates. However, in < 1% of cases, there can be injury to nearby bowel or bladder, pelvic infection or bleeding that requires a blood transfusion.
- Procedure-associated complications: By far the most common type of complications, these include multiple births, possibly higher birth defect rates and concerns about an increase in imprinting disorders in offspring. The high incidence of multiple gestations is the major concern with ART procedures. Multiple pregnancies occur up to ~35% of ART pregnancies, compared to 1 to 2% of naturally conceived pregnancies. Multiple gestations are associated with lower birth weights, higher overall complication rates and increased learning disabilities relative to singletons.

There are also several concerns regarding the health of offspring obtained after IVF-ICSI. Although the overall rates of major birth defects associated with IVF and ICSI (3.3%) are similar to birth defect rates observed in children conceived after intercourse or IVF alone, there is concern about the risks of sex chromosome anomalies and other specific malformations, including hypospadias, in IVF-ICSI offspring. Two other concerns with IVF-ICSI are:
(1) Since IVF and ICSI may eliminate many natural-selection barriers that exist during natural fertilization, genetic defects that caused the infertility are expected to be passed on to offspring unabated.
(2) Recent data show that offspring born to infertile couples with this technique have a 4-fold higher rate (0.8%) of sex chromosomal anomalies than do children who are naturally conceived. Even more recently, concerns have arisen regarding whether rare imprinting diseases such as Beckwith-Weideman Syndrome and Anglemann Syndrome are increased in children conceived with ICSI. It is also controversial whether subtle development defects and delays exist in ART children.
How ART compare with other treatments for male infertility.
Meet with Dr. Turek
Dr. Turek is one of the most celebrated experts when it comes to male infertility and ART. Having great experience with, and even pioneering, several fertility-related procedures used with ART, he has a long track record of successfully helping couples who have fertility issues to finally conceive. He is committed to finding solutions for patients in Los Angeles and San Francisco that result in pregnancy. Please schedule a consultation online or call 1-888-TUREKMD.









